- Schedule. Mon - Sat 9.00 - 18.00
Quality& Certifications
Safest and most dependable choice
HtsinPrecison is AS 9100 D / ISO 9001: 2015 certified and additional certifications are available through our manufacturing partners.
Understanding ISO 9001 & AS9100 Certifications in Machining
Certifications are no longer an optional layer in modern precision manufacturing. In high-spec industries, they are essential benchmarks for quality, traceability, and operational excellence. ISO 9001 and AS9100 are globally recognized certifications that signal a manufacturer’s commitment to stringent quality management in CNC machining.
The importance of Certifications in Machining has grown significantly over the past decade. With supply chains tightening and product tolerances shrinking, original equipment manufacturers (OEMs) prioritize certified suppliers who can demonstrate documented quality control, process consistency, and risk mitigation. According to the International Aerospace Quality Group, over 80% of global aerospace companies require AS9100 certification from CNC suppliers.
Whether it’s aircraft brackets, medical implants, or EV motor housings, customers now demand more than just precision—they require proof of repeatability, reliability, and compliance. That’s where ISO 9001 and AS9100 come in.
What is the Difference Between ISO 9001 and AS9100?
While both certifications in Machining reinforce quality assurance in CNC machining, their frameworks serve different operational risk levels. ISO 9001 provides foundational quality management applicable across sectors, while AS9100 embeds specific controls for aerospace, defense, and space systems, where failure is not an option. Below is a detailed technical breakdown:
| Technical Category | ISO 9001 | AS9100 |
|---|---|---|
| Applicability Scope | General manufacturing industries | Aerospace, defense, and high-reliability sectors |
| Design and Configuration Control | May include design clause if applicable; often excluded for contract-only manufacturing | Mandatory design clause with detailed configuration management per project scope |
| Risk Management Methodology | Risk is identified but not embedded in all processes | Formal risk-based thinking applied across planning, production, supply chain, and customer interface |
| Product Safety Requirements | Not explicitly required under the standard | Mandatory product safety protocols integrated into design, production, and post-delivery activities |
| Process Documentation & Record Retention | Basic document control; minimal retention period based on contract | Detailed document control with serialized traceability, extended record retention (up to 10 years or more) |
| Supplier Quality Management | Supplier selection based on general quality criteria | Formal supplier development, qualification, risk assessment, and scheduled supplier performance audits |
| First Article Inspection (FAI) | Not required; may be customer-specified | Mandatory per AS9102; all dimensional, material, and process variables must be validated before full-scale production |
| Nonconformance Management | Basic corrective action system | Requires formal root cause analysis (RCA), systemic corrective and preventive actions (CAPA), and effectiveness review |
| Change Control | Informal may vary by project | Engineering change orders (ECOs) must follow structured configuration and documentation change approval protocols |
| Counterfeit Part Prevention | Not addressed | The mandatory program required to identify, control, and prevent counterfeit or unapproved components/materials |
| Process Validation & Verification | General requirement for process validation | Mandatory verification for special processes (e.g., welding, NDT, plating) that cannot be verified by subsequent inspection |
| Customer Satisfaction Monitoring | Encouraged; tracked via complaints and feedback | Structured, data-driven approach for measuring customer satisfaction, on-time delivery, and complaint resolution |

01
Quality testing laboratory
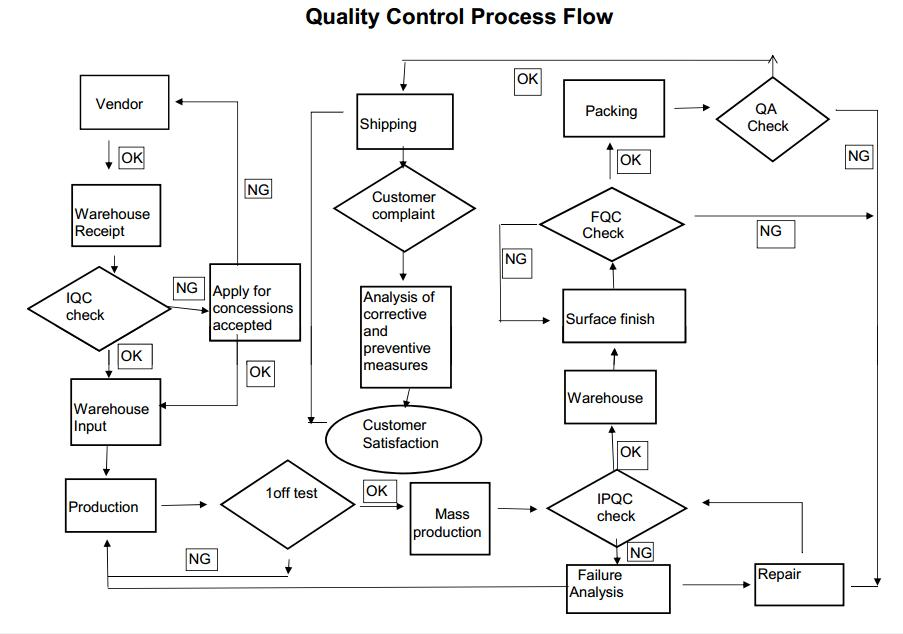
Our quality process is run through the whole projects from RFQs to production shipment.
Two independent reviews of the purchase order is where our QA begins, determining that there are no questions or conflicts regarding dimensions, material, quantities, or delivery dates.
Then reviewed by experienced personnel involved in the set up and production and individual inspection reports are made for each operation that is required to produce the part.
All special quality needs and instructions are documented and inspection intervals are then assigned based on tolerances, quantities or complexity of the part.
We minimize risk by tracking and analyzing every step of our manufacturing process to minimize part to part variation, and assure consistent, reliable quality for every part, every time.
02
Highly experienced htsin online inspector
Consistent quality control, from part to part, product to project, we pay attention to detail, problem-solving, researching new materials and processes, investing in technology, building and sustaining a team of professional rapid manufacturing.
Design for Manufacturing (DFM) review for all of your projects.
Production capacity and production plan review (PMC).
Incoming raw materials inspection.
Samples and in-process inspection (IPQC).
Control of nonconforming product and the implementation of corrective and preventive actions.
Final inspection and testing reports and certifications as required(OQC).


03
Advanced Inspection Equipment
ScanTech PRINCE775 Handheld 3D Scanner: Laser source – 14+1 red laser crosses / 5 blue parallel laser lines; Effective work range 275 mm×250 mm / 200 mm×200 mm; Accuracy Up to 0.03mm.
KEYENCE lM 8010 lmage Dimension Measurement System (Optical Comparator): 200×200(mm), With a 20-megapixel CMOS sensor, Measure 300 features in seconds, Measurement accuracy ±3.9μm (Wide-field ), ±2.0μm (High-precision).
TL4030PC Optical 2D Vision Image Measurement Machine: 400x300x200(mm), Measurement accuracy EI (X/Y) ≤ (3.5+L/200) μm.
Mitutoyo SURFTEST SJ-210 Surface Roughness Tester: Measuring range 360µm ( -200µm – +160µm ); Range / Resolution 360µm / 0.02µm, 100µm / 0.006µm, 25µm / 0.002µm.
TESA-HITE 700 Digimatic Heitht Gauge: Measuring range 715 mm; Resolution 0.0001 / 0.001 / 0.01 mm; Max. perm. Errors 2.5+4L µm.
At Htsin traceability is integrated into every machining process through comprehensive lot control and tracking systems. Using barcoding and RFID technology, we ensure every part is linked to raw material sources, tool configurations, machine settings, and inspection records. This meets the rigorous traceability standards of ISO 9001 and AS9100, which are essential for critical industries such as aerospace, defense, and medical devices.
Htsin uses state-of-the-art coordinate measuring machines (CMM), laser scanning, and vision systems to perform dimensional and surface finish inspections on high-precision aerospace parts. Our AS9100-compliant inspection process includes verifying features down to micrometer-level tolerances and implementing First Article Inspections (FAI) following AS9102 to ensure strict conformance to aerospace standards.
Htsin adheres to a stringent quality management system per ISO 9001 in CNC machining for medical devices. We implement Process Control Plans (PCPs), monitor critical-to-quality (CTQ) parameters such as dimensional accuracy and surface finish, and provide full documentation for traceability. All production processes are validated with machine capability studies (Cpk), ensuring that parts like bone fixation plates and diagnostic components meet FDA and EU MDR standards.
Under AS9100, Htsin employs a detailed nonconformance management system that includes root cause analysis (RCA) implementing corrective and preventive actions (CAPA). We utilize statistical tools like Pareto Analysis and Fishbone Diagrams to identify the underlying causes of nonconformance. Our corrective actions are thoroughly documented, ensuring compliance with AS9100 standards and preventing recurrence, which is critical in sectors like defense and aerospace.
Htsin utilizes fully automated CNC machining systems with integrated Real-Time Data Monitoring (RTDM) and in-process inspection to ensure consistent quality. These systems are calibrated to maintain exact tolerances, significantly reducing human error. Automated feedback loops, combined with Statistical Process Control (SPC), guarantee that all parts, such as missile interface hardware and aircraft brackets, meet AS9100 standards for precision and performance.
Through advanced material traceability systems, Frigate manages high-strength materials like Inconel, titanium, and aerospace alloys. Each batch of raw material is certified to ASTM standards, and the machining process is meticulously controlled with material certificates of conformance (MCOCs). We implement heat treatment and stress relief processes as required to maintain the integrity of these critical materials, ensuring compliance with ISO 9001 and AS9100.
At Htsin, risk management is embedded into every phase of the machining process. We conduct Failure Mode Effects Analysis (FMEA) and Risk Assessment Reports at the design and production stages. This proactive approach ensures that parts like aerospace turbine housings or defense missile components are produced with minimal risk of failure. This risk-driven process is fully documented to comply with ISO 9001 and AS9100.
Htsin employs a robust design revision control system that uses Engineering Change Orders (ECOs) to track every change. Under AS9100, these changes are reviewed for impact on part performance, material properties, and overall process flow. The revision history is fully documented and auditable, ensuring that customer-driven changes are seamlessly integrated without compromising compliance with ISO 9001 or AS9100.
Htsin utilizes Lean Manufacturing principles and automated in-line inspection systems for high-volume automotive machining. Every part undergoes Statistical Process Control (SPC) to maintain dimensional accuracy and surface finish, ensuring consistency. Automated setup changes and process optimization minimize downtime, and all processes are compliant with ISO 9001 to ensure that parts like powertrain brackets or battery trays meet high-volume quality standards.
In medical machining, Htsin employs controlled environments for critical processes, ensuring compliance with ISO 9001 and AS9100. All medical components, such as surgical guides or diagnostic fixtures, are fabricated using validated processes and undergo rigorous validation protocols, including dimensional CMM verification. Additionally, we maintain full regulatory documentation to meet FDA, CE, and ISO 13485 standards, ensuring that each component fully complies with all relevant medical regulations.
